The World Health Organization (WHO) has issued a global alert on falsified semaglutide products that are used for the treatment of type 2 diabetes and obesity in some countries.
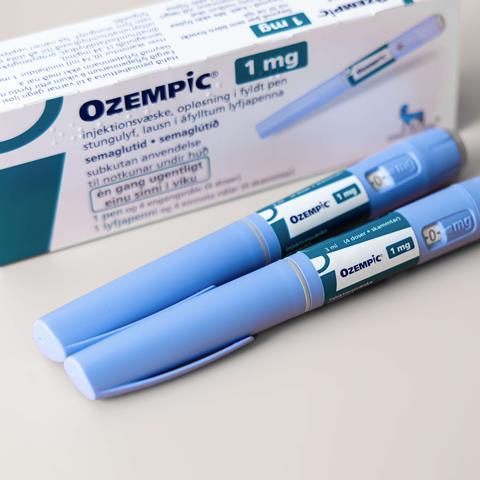
The alert addresses three falsified batches of the product under the brand name Ozempic which have been detected in Brazil, the UK and Northern Ireland in October 2023, and in the US in December 2023.
The falsified pens contain incorrect batch and unique serial numbers or, in some instances, genuine batch numbers on falsified pens.
The organisation said in a press release that it has been observing increasing numbers of reports of falsified semaglutide products in all geographical regions since 2022 through its Global Surveillance and Monitoring System (GSMS). This is the first official notice issued by the WHO after confirmation of some reports.
The organisation has warned that the falsified pens could pose serious risks to people’s health and has advised people to examine their pens’ features by looking for spelling mistakes and assessing the label quality.
It also urged people to source semaglutide products from licensed physicians under prescription and to avoid buying from unfamiliar or unverified sources.
“[The] WHO advises healthcare professionals, regulatory authorities and the public [to] be aware of these falsified batches of medicines,” said Dr Yukiko Nakatani, the WHO’s Assistant Director-General for Access to Medicines and Health Products.
“We call on stakeholders to stop any usage of suspicious medicines and report to relevant authorities,” he added.
Further reading
The WHO: Medical Product Alert N°2/2024: Falsified OZEMPIC (semaglutide)
Regulatory Rapporteur: EU patients warned about falsified Ozempic medication
Regulatory Rapporteur: Off-label prescription of diabetes drug causes shortage