All Transfusion-dependent β-thalassemia articles
-
 Journal
JournalThe first approval of a CRISPR-Cas9 gene editing medicinal product in the European Union: Casgevy
Regulatory Rapporteur May 2024 | Volume 21 | No.5 Abstract Patients with transfusion-dependent β-thalassemia and severe sickle cell disease suffer the burden of repeated blood transfusions and vaso-occlusive crises, respectively. Casgevy (exagamglogene autotemcel) consists of autologous CD34+ haematopoietic stem ...
-
 Industry news
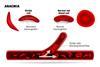
Industry newsCRISPR tool used to treat sickle-cell and β-thalassemia
Casgevy is the first gene therapy that uses the CRISPR/Cas9 gene-editing tool to be approved by the UK MHRA.



















